Access the PDF Version Here: Soft Spots
Access the Service Industry News Article Reprint Here: SIN 15 Jan 2003 Spotting Reprint
Plaster Soft Spots
Quick Facts:
What does it look like?
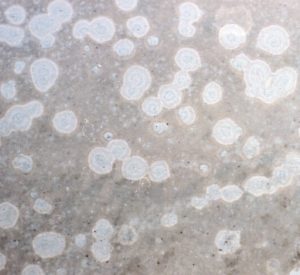
Generally round spots about the size of a pencil eraser to a dime, and usually much whiter than surrounding areas of the plaster (both white and colored plaster). May become tan or aqua over time by absorbing dissolved iron or copper. May follow the edge of tile, highlight plaster cracks, or follow a troweled radius, or outline low spots in the plaster.
Surrounded by smooth, dense, intact plaster. Although aggregate (sand) is visible in the spotted area itself, the surrounding area is smooth cement paste.
Even though the spot shows aggregate, the spot is relatively smooth.
How does it happen?
Over-accelerated (and thus porous) plaster is late hard troweled, disturbing aggregate at surface. Water-troweling allows water to penetrate the paste-to-aggregate interface. Over time, water in the filled pool leaches soft paste from around the surface aggregate, and continues leaching in a pattern radiating from the point of origin.
Can it be fixed? How?
No – this is a permanent, internal to the surface effect that is non-structural but non-removable.
Can it be prevented in the future?
Spotting can be prevented by reducing or eliminating calcium chloride accelerator use, proper timing of troweling operations, and eliminating wet-finishing techniques.
Whose fault was it?
Plastering crew
Plaster soft spots are surface blemishes that are caused by a combination of plastering errors. These include the over-use of calcium chloride set accelerant, and (as subsequent coping techniques) late over-troweling and adding water to the surface during late troweling.
 This phenomenon has been referred to by some in the past as “spot etching” but this is a misnomer because it is not an etching process, but rather one of leaching. Leaching is the capability of water (any water, regardless of its chemical characteristics) to act as a solvent – i.e., to dissolve things. Water is popularly called “the universal solvent” because it can solubilize materials just by being water. Of course, some soft “soluble” materials will dissolve in seconds, while other materials may require days, weeks, months, years – even centuries or millennia to dissolve in normal water. Etching, on the other hand, describes a chemical attack which occurs specifically because of the aggressive qualities of the water. In swimming pools, aggressive water is generally water that has low pH, low alkalinity, low hardness, high dissolved solids (especially as salt), etc., or a combination of the above, which makes it aggressive to plaster – a highly alkaline (the opposite of acid) material.
This phenomenon has been referred to by some in the past as “spot etching” but this is a misnomer because it is not an etching process, but rather one of leaching. Leaching is the capability of water (any water, regardless of its chemical characteristics) to act as a solvent – i.e., to dissolve things. Water is popularly called “the universal solvent” because it can solubilize materials just by being water. Of course, some soft “soluble” materials will dissolve in seconds, while other materials may require days, weeks, months, years – even centuries or millennia to dissolve in normal water. Etching, on the other hand, describes a chemical attack which occurs specifically because of the aggressive qualities of the water. In swimming pools, aggressive water is generally water that has low pH, low alkalinity, low hardness, high dissolved solids (especially as salt), etc., or a combination of the above, which makes it aggressive to plaster – a highly alkaline (the opposite of acid) material.
The mechanical sequence of soft-spotting plaster is usually as follows:
Excess calcium chloride is added to the plaster mix. Calcium chloride is often used as a set accelerant, but it must be used sparingly, if at all. Calcium chloride discolors concrete (including plaster) at levels as low as 1%. It creates shrinkage and excess porosity at levels relational to the amount added, as well as relative to environmental factors including both ambient temperature and plaster mix temperature. There is an accepted industry standard to not exceed 2% calcium chloride addition, although this is a maximum for challenging conditions (such as cold weather plastering). Less or no calcium chloride should be used in warmer or hot temperature plastering. Non-chloride accelerators, which do not have the shrinkage and discoloration issues that chloride-based accelerants have, are available as an alternative.
Calcium chloride is measured in plaster by its percentage relative to the cement component by weight. It is reported as “percent calcium chloride as dihydrate by weight to cement.” Petrographic analysis of plaster that developed the unsightly spots has shown levels ranging from 2 to 5%, even though some of these pools were plastered in warm or even hot conditions.
If a pool is setting up too fast, plastering finishers must over-trowel, and trowel later in the setting process than normal. This over-troweling, performed to quickly produce the smooth finish required as the plaster sets too fast, may be done late enough in the process that aggregate, (in this case, sand) at the surface of the plaster is scraped and vibrated in its paste bed, creating a weak, broken paste-to-aggregate interface. This interface, called an inter-transitional zone, then provides an entry path for water. 
It is against accepted cement/concrete trade practice to trowel bleed water (water rising to the top of a concrete mix) back into the paste mass. Instead, bleed water is supposed to evaporate from the surface between trowel passes. Pushing bleed water back into the paste results in a weak subsurface that can be exploited by wear, pressure, water, and even age. Unfortunately, it has become common by some in the plaster finishing industry to actually apply water to the surface via brushes or sponges and troweling that water into the surface. Not only does this practice result in a weaker surface, but when aggregate at surface has been disturbed as described above, this applied water penetrates around the aggregate/paste boundary and creates a soluble, weak crystalline matrix of calcium hydroxide.
When such a pool is filled with water, over the course of days, weeks and months water leaches the weak hydroxide from around the surface-exposed aggregate, and then continues to leach laterally in a pattern radiating outward through the chloride- and water-generated porous surface paste, thus creating the distinctive halo of a plaster soft spot with a dot of sand in the middle.
Although some plaster soft spots may be visible even as the pool fills, by far the most spots become visible and objectionable in the 2 to 6 month period after plastering.
 Spots are significantly more porous than the surrounding paste, and are therefore the first to absorb heavy metals, becoming tan or turquoise as a result. Some “spot etch removal” formulations are simply chelating agents which remove the copper or iron. The spot is then white-on-white again, and therefore less noticeable. Of course, spots on colored plaster are whiter, and this solution does not work.
Spots are significantly more porous than the surrounding paste, and are therefore the first to absorb heavy metals, becoming tan or turquoise as a result. Some “spot etch removal” formulations are simply chelating agents which remove the copper or iron. The spot is then white-on-white again, and therefore less noticeable. Of course, spots on colored plaster are whiter, and this solution does not work.
The combination of factors contributing to surface spotting in concrete have been identified by Portland Cement Association researchers, and they have been tied to pool plaster specifically by forensic cement petrographers.
A “Mechanical Model” of How Spots are Formed
 This is a scanning electron microscope image of the cross-section of a soft spot. The black area at the top is the water, the large gray objects are individual grains of sand.
This is a scanning electron microscope image of the cross-section of a soft spot. The black area at the top is the water, the large gray objects are individual grains of sand.
 Now we have turned it into a color drawing for illustration.
Now we have turned it into a color drawing for illustration.
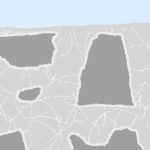
 When too much set accelerator is added to plaster, the cement paste “shrinks” and creates crazing and micro-porosity, like this mud flat.
When too much set accelerator is added to plaster, the cement paste “shrinks” and creates crazing and micro-porosity, like this mud flat.
 The sand doesn’t shrink – just the cement paste around it shrinks and pulls away from the sand.
The sand doesn’t shrink – just the cement paste around it shrinks and pulls away from the sand.
 Because of the shrinkage, the cement paste is also full of microcracks in an interlaced network.
Because of the shrinkage, the cement paste is also full of microcracks in an interlaced network.
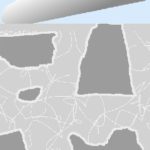 Since the plaster is drying so fast, the plastering crew needs to “knock it down” smooth, sometimes with late, excessive hard troweling.
Since the plaster is drying so fast, the plastering crew needs to “knock it down” smooth, sometimes with late, excessive hard troweling.
 As the trowel hits aggregate at surface it can “jiggle” the sand in its loose paste bed, creating a bigger, looser pocket in the paste.
As the trowel hits aggregate at surface it can “jiggle” the sand in its loose paste bed, creating a bigger, looser pocket in the paste.
 Sometimes the plasterer also wets the surface with water – which is a no-no, but helps him get the surface flat and smooth.
Sometimes the plasterer also wets the surface with water – which is a no-no, but helps him get the surface flat and smooth.
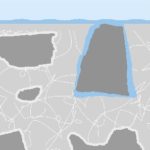 As this water is forced into the surface, it surrounds the aggregate.
As this water is forced into the surface, it surrounds the aggregate.
 As the water dries, it leaves behind a loose latticework of crystalline calcium hydroxide in its place.
As the water dries, it leaves behind a loose latticework of crystalline calcium hydroxide in its place.
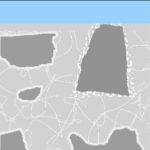 As the pool is later filled…
As the pool is later filled…
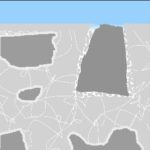 …water penetrates the area around the sand grain and leaches the weak material away. This leaching is due to the solvency of even perfectly balanced water – although it happens faster because of the inferior, compromised paste.
…water penetrates the area around the sand grain and leaches the weak material away. This leaching is due to the solvency of even perfectly balanced water – although it happens faster because of the inferior, compromised paste.
 Leaching then spreads laterally through the micro-cracking fissures, forming the characteristic round spot. This can continue until many spots coalesce into bigger spots.
Leaching then spreads laterally through the micro-cracking fissures, forming the characteristic round spot. This can continue until many spots coalesce into bigger spots.
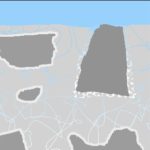 Aggressive water will make the process happen faster and maybe deeper, but the defect ensures that the spot will form even in perfectly balanced water.
Aggressive water will make the process happen faster and maybe deeper, but the defect ensures that the spot will form even in perfectly balanced water.
